DOI:
https://doi.org/10.22516/25007440.535Palavras-chave:
SARS-CoV, SARS-CoV-2, MERS-CoV, hígado, trasplante de hígado, inmunosupresiónResumo
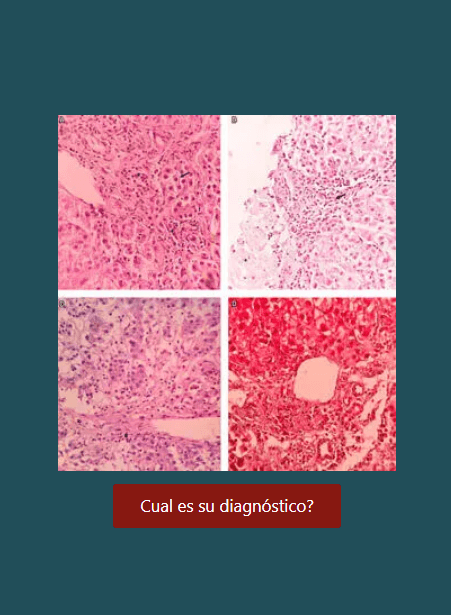
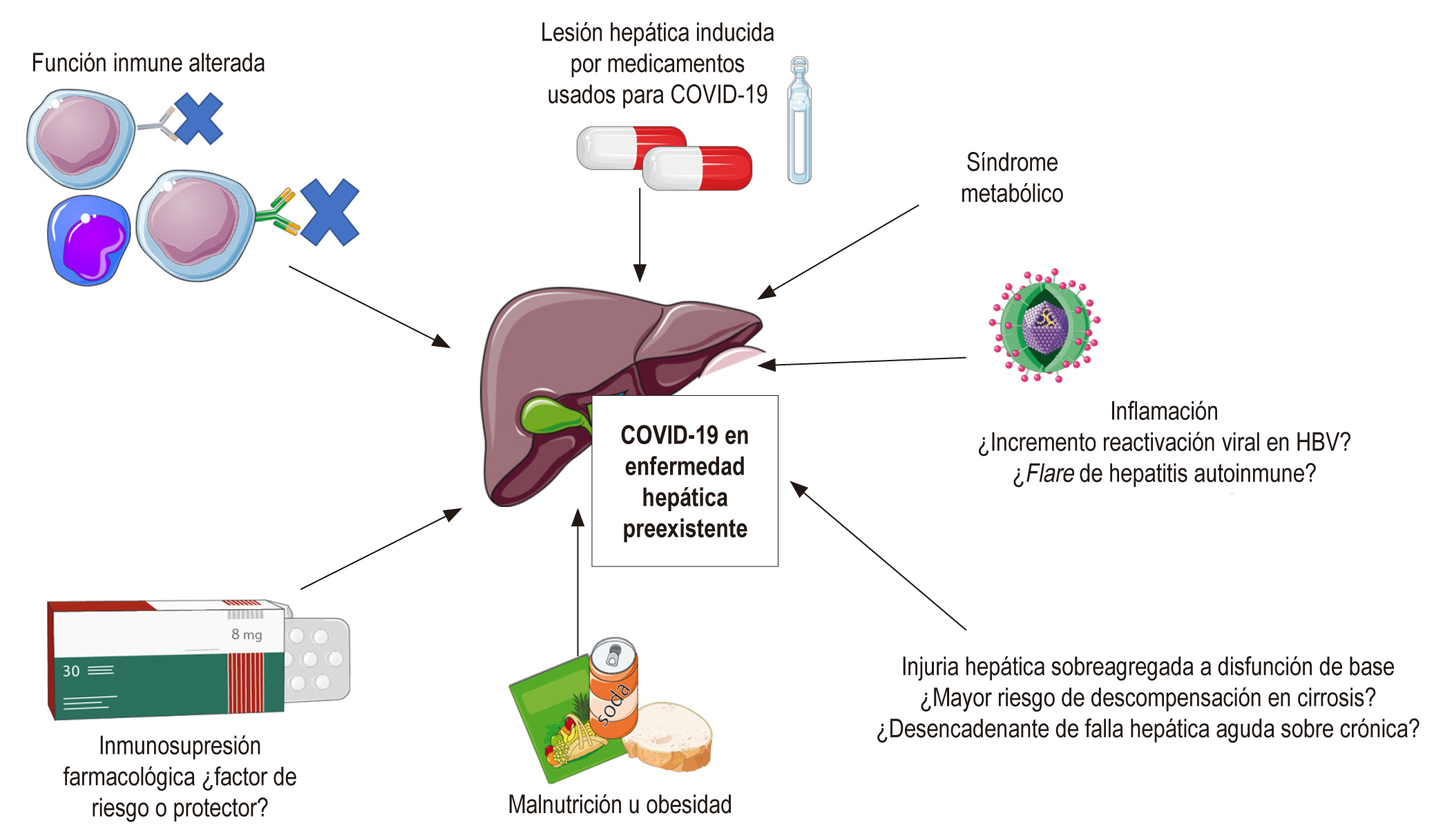
La infección generada por el coronavirus, denominado SARS-CoV-2, llamada coronavirus disease 2019 (COVID-19), surgió en China a finales de diciembre de 2019. Actualmente ha sido categorizada como una pandemia por la Organización Mundial de la Salud (OMS). Se han documentado alteraciones de pruebas hepáticas, sin embargo, los estudios se han enfocado en los efectos cardíacos, pulmonares y renales de esta infección. La alteración de pruebas hepáticas en el contexto de COVID-19 puede ser consecuencia de hepatitis viral, toxicidad farmacológica, inflamación o choque. También se considera como un marcador de pronóstico y gravedad de la enfermedad. El impacto de la infección por SARS-CoV-2 en pacientes con enfermedad hepática preexistente o receptores de trasplante hepático no es claro, y se plantean distintas hipótesis sobre mayor o menor riesgo de enfermedad grave y de descompensación de la enfermedad de base.
Downloads
Referências
WHO [Internet]. WHO; 2020 [actualización 11 de marzo de 2020; acceso 24 de abril de 2020]. Director-General’s opening remarks at the media briefing on COVID-19 - 11 March 2020. Disponible en: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020
European Center for Disease Prevention and Control [Internet]. ECDC; 2019. [actualización 2 de abril de 2020; acceso 24 de abril de 2020]. Situation update worldwide, as of 02 April 2020. Disponible en: https://www.ecdc.europa.eu/en/geographical-distribution-2019-ncov-cases
Instituto Nacional de Salud [Internet]. INS; 2020 [acceso 24 de abril de 2020]. COVID-19 Colombia - Reporte 01-04-2020-Corte 1pm. Disponible en: https://www.ins.gov.co/Noticias/Paginas/Coronavirus.aspx
Chau TN, Lee KC, Yao H, et al. SARS-associated viral hepatitis caused by a novel coronavirus: report of three cases. Hepatology. 2004; 39:302-10. https://doi.org/10.1002/hep.20111
Alsaad KO, Hajeer AH, Al Balwi M, et al. Histopathology of Middle East respiratory syndrome coronavirus (MERS-CoV) infection - Clinicopathological and ultrastructural study. Histopathology. 2018; 78:516-24. https://doi.org/10.1111/his.13379
Xu L, Liu J, Lu M, Yang D, Zheng X. Liver injury during highly pathogenic human coronavirus infections. 2020. https://doi.org/10.1111/liv.14433
Li W, moor MJ, Vasilieva N, et al. Angiotensin-converting enzime 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426(6965):450-54. https://doi.org/10.1038/nature02145
Hamming I, Timens W, Bulthuis M, et al. Tissude distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Patho. 2004;203(2):631-37. https://doi.org/10.1002/path.1570
Tan YJ, Fielding BC, Goh PY, et al. Overexpression of 7a, a protein specifically encoded by thesever acute respiratory syndrome coronavirus, induces apoptosis via a caspase-depedent pathway. J Virol. 2004;78(24):14043-47. https://doi.org/10.1128/JVI.78.24.14043-14047.2004
Lu J, Zhao J, Li N, et al. Ultrastructurte pathology of all organs in sever acute respiratory syndrome. Chinese Journal of Diagnostic Pathology. 2003;04:72-7.
Saad M, Omrani AS, Baig K, et al. Clinical aspects and outcomes of 70 patients with Middle East respiratory syndrome coronavirus infection: a single-center experience in Saudi Arabia. Int J Infect Dis. 2014;29:301-06. https://doi.org/10.1016/j.ijid.2014.09.003
Arabi YM, Arifi AA, Balkhy HH, et al. Clinical course and outcomes of critically ill patients with Middle East respiratory syndrome coronavirus infection. Ann Intern Med. 2014:160(6):389-97. https://doi.org/10.7326/M13-2486
Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020. https://doi.org/10.1016/S2468-1253(20)30057-1
Chen N, Zhou M, Qu J, Gong F, Han Y, Qiu Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020; 395:507-13. https://doi.org/10.1016/S0140-6736(20)30211-7
Wu C, Chen Z, Cai Y, Xia J, Zhou X, Xu S, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med. https//doi.org/10.1001/jamainternmed.2020.O994
Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054-62. https://doi.org/10.1016/S0140-6736(20)30566-3
Wang D, Hu B, Zhu F, Liu X, Zhang J, Wang B, et al. Clinical characteristics of 138 patients with 2019 novel coronavirus-infected pneumonia in Wuhan-China. JAMA. 2020. https://doi.org/10.10001/jama.2020.1585.
Rismanbaf A, Zarei S. Liver and Kidney Injuries in COVID-19 and Their Effects on DrugTherapy; a Letter to Editor. Archives of Academic Emergency Medicine. 2020;8(1):e17.
Yang Z, Xu M, Yi JQ, et al. Clinical characteristics and mechanism of liver damage in patients with severe acute respiratory syndrome. Hepatobiliary Pancreat Dis Int. 2005;4(1):60-63.
Bangash MN, Patel J, Parekh D. COVID-19 and the liver: little cause for concern. Lancet Gastroenterol Hepatol. 2020. https://doi.org/10.1016/S2468-1253(20)30084-4
Zhang B, Zhou X, Qiu Y, et al. Clinical characteristics of 82 death cases with COVID-19. medRxiv. 2020. https://doi.org/10.1101/2020.02.26.20028191
Huang Y, Gao Z. Study of the relationship SARS and hepatitis virus B. Chin J Clini Hepatol. 2003;6:342-343.
Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ. COVID-19: consider cytokine storm syndromes and immunosuppression. The Lancet. 2020. https://doi.org/10.1016/S0140-6736(20)30628-0
Xiao Y, Pan H, She Q, Wang F, Chen M. Prevention of SARS-CoV-2 infection in patients with decompensated cirrhosis. Lancet Gastroenterol Hepatol. 2020. https://doi.org/10.1016/S2468-1253(20)30080-7
Boettler, T. Newsome PN, Mondelli MU, Maticic M, Cordero E, Cornberg M, Berg T. Care of patients with liver disease during the COVID-19 pandemic: EASL-ESCMID Position Paper. JHEP Reports. https://doi.org/10.1016/j.jhepr.2020.100113
American Association For The Study of Liver Disease [Internet]. AASLD; 2020 [actualización 16 de abril de 2020; acceso 24 de abril de 2020]. Clinical insights for hepatology and liver transplant providers during the COVID-19 Pandemic. Disponible en: https://www.aasld.org/sites/default/files/2020-03/AASLD-COVID19-ClinicalInsights-3.23.2020-FINAL-v2.pdf
D’Antiga L. Coronaviruses and immunosuppressed patients. The facts during the thrid epidemic. Liver Transpl. 2020. https://doi.org/10.1002/lt.25756
Guo YR, Cao QD, Hong ZS, Tan YY, Chen SD, Jin HJ, et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak - an update on the status. Military Medical Research. 2020;7(1):11. https://doi.org/10.1186/s40779-020-00240-0
Kumar D, Tellier R, Draker R, Levy G, Humar A. Severe Acute Respiratory Syndrome (SARS) in a Transplant Recipient and Guidelines for Donor SARS Screening. American Journal of Transplantation. 2003;3(8):977-81. https://doi.org/10.1034/j.1600-6143.2003.00197.x
Downloads
Arquivos adicionais
Publicado
Como Citar
Edição
Seção
Licença
Aquellos autores/as que tengan publicaciones con esta revista, aceptan los términos siguientes:
Los autores/as ceden sus derechos de autor y garantizarán a la revista el derecho de primera publicación de su obra, el cuál estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons que permite a terceros compartir la obra siempre que se indique su autor y su primera publicación en esta revista.
Los contenidos están protegidos bajo una licencia de Creative Commons Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional.


| Métricas do artigo | |
|---|---|
| Vistas abstratas | |
| Visualizações da cozinha | |
| Visualizações de PDF | |
| Visualizações em HTML | |
| Outras visualizações | |