Hepatocellular carcinoma: A real-life experience in a specialized center in Bogotá, Colombia
DOI:
https://doi.org/10.22516/25007440.823Keywords:
Hepatocellular carcinoma, Real life, Survival, SorafenibAbstract
Introduction: Hepatocellular carcinoma (HCC) is the most frequent malignant primary liver tumor globally. In 2018, it ranked sixth and represented the fourth cause of death from cancer; the five-year overall survival is 18 %. Most cases of HCC develop in patients with cirrhosis of any etiology, especially because of hepatitis B and C viruses, alcohol, and recently nonalcoholic steatohepatitis (NASH).
Aim: To analyze the clinical characteristics, diagnostic methods, treatments, prognostic variables, and survival.
Materials and methods: This retrospective descriptive study was conducted on a cohort of patients diagnosed with cirrhosis and treated between January 2011 and December 2020 at a health care center in Bogotá. The diagnosis of HCC was confirmed radiologically or by biopsy. We analyzed the information descriptively with absolute frequency measures in the case of categorical variables. For continuous variables, the information was summarized with measures of central tendency (mean or median) and their relevant measures of dispersion.
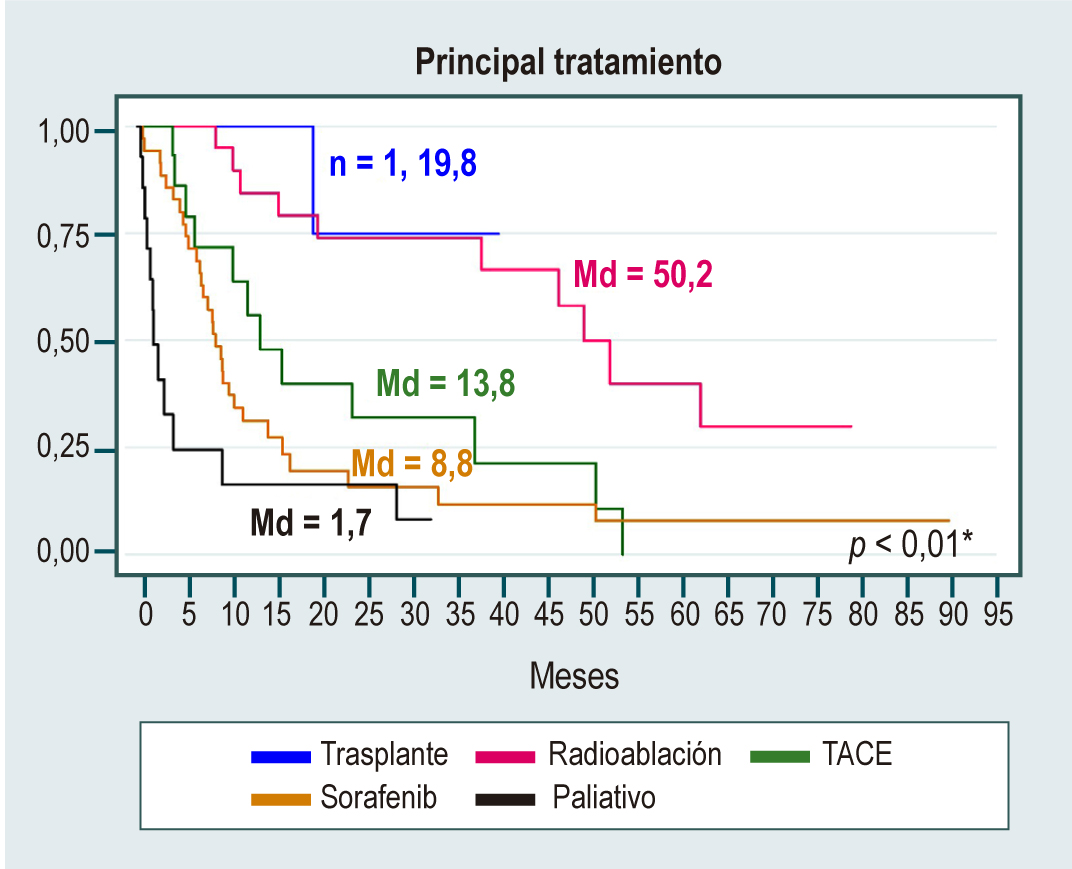
Results: We included 152 patients diagnosed with HCC, with a mean age of 69.4 years; 51.3 % were men. The leading cause of HCC was nonalcoholic fatty liver disease (NAFLD), which accounted for almost a third of cases (32 %); other causes were alcohol (15 %) and hepatitis C virus (14 %). The median manifestation of the tumor was two nodules with a size close to 4 cm. Besides, 35 % of patients had a BCLC (Barcelona Clinic Liver Cancer) stage with curative options, and 25 % received curative treatment options. The first-line systemic therapy used in this cohort was sorafenib®, used in 35 patients (33.7 %). Survival curves showed that women, Child-Pugh class A, and BCLC stage 0 had higher median survival. Multivariate analysis showed a higher risk of death for males (hazard ratio [HR]: 2.16; confidence interval [CI]: 1.24–3.76), Child-Pugh class B (HR: 2.14; CI 1.16–3.95), and Child-Pugh class C (HR: 7.52; CI 2.88–19.57).
Conclusions: NAFLD is the leading cause of HCC in this cohort. A third of patients are diagnosed in early BCLC stages with a curative treatment option, and 25 % are treated with curative therapies. Sorafenib was the first-line therapy in advanced HCC. Overall survival after diagnosis of HCC remains low, being necessary to join forces in the follow-up of patients with cirrhosis to improve these outcomes.
Downloads
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394-424. https://doi.org/10.3322/caac.21492
Global Burden of Disease Liver Cancer Collaboration, Akinyemiju T, Abera S, Ahmed M, Alam N, Alemayohu MA, et al. The Burden of Primary Liver Cancer and Underlying Etiologies From 1990 to 2015 at the Global, Regional, and National Level: Results From the Global Burden of Disease Study 2015. JAMA Oncol. 2017;3(12):1683-1691. https://doi.org/10.1001/jamaoncol.2017.3055
Jemal A, Ward EM, Johnson CJ, Cronin KA, Ma J, Ryerson B, et al. Annual Report to the Nation on the Status of Cancer, 1975-2014, Featuring Survival. J Natl Cancer Inst. 2017;109(9):djx030. https://doi.org/10.1093/jnci/djx030
Kulik L, El-Serag HB. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology. 2019;156(2):477-491.e1. https://doi.org/10.1053/j.gastro.2018.08.065
Pardo C, de Vries E, Buitrago L, Gamboa O. Atlas de mortalidad por cáncer en Colombia. 4.a edición. Bogotá D. C.: Instituto Nacional de Cancerología; 2017. p. 124.
Fattovich G, Stroffolini T, Zagni I, Donato F. Hepatocellular carcinoma in cirrhosis: incidence and risk factors. Gastroenterology. 2004;127(5 Suppl 1):S35-50. https://doi.org/10.1053/j.gastro.2004.09.014
Kanwal F, Hoang T, Kramer JR, Asch SM, Goetz MB, Zeringue A, et al. Increasing prevalence of HCC and cirrhosis in patients with chronic hepatitis C virus infection. Gastroenterology. 2011;140(4):1182-1188.e1. https://doi.org/10.1053/j.gastro.2010.12.032
El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118-27. https://doi.org/10.1056/NEJMra1001683
Sangiovanni A, Prati GM, Fasani P, Ronchi G, Romeo R, Manini M, et al. The natural history of compensated cirrhosis due to hepatitis C virus: A 17-year cohort study of 214 patients. Hepatology. 2006;43(6):1303-10. https://doi.org/10.1002/hep.21176
Suresh D, Srinivas AN, Kumar DP. Etiology of Hepatocellular Carcinoma: Special Focus on Fatty Liver Disease. Front Oncol. 2020;10:601710. https://doi.org/10.3389/fonc.2020.601710
Makarova-Rusher OV, Altekruse SF, McNeel TS, Ulahannan S, Duffy AG, Graubard BI, et al. Population attributable fractions of risk factors for hepatocellular carcinoma in the United States. Cancer. 2016;122(11):1757-65. https://doi.org/10.1002/cncr.29971
Bruix J, Sherman M, Llovet JM, Beaugrand M, Lencioni R, Burroughs AK, et al. Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol. 2001;35(3):421-30. https://doi.org/10.1016/s0168-8278(01)00130-1
Bruix J, Sherman M; Practice Guidelines Committee, American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma. Hepatology. 2005;42(5):1208-36. https://doi.org/10.1002/hep.20933
Matsui O, Kobayashi S, Sanada J, Kouda W, Ryu Y, Kozaka K, et al. Hepatocelluar nodules in liver cirrhosis: hemodynamic evaluation (angiography-assisted CT) with special reference to multi-step hepatocarcinogenesis. Abdom Imaging. 2011;36(3):264-72. https://doi.org/10.1007/s00261-011-9685-1
Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, et al. Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68(2):723-750. https://doi.org/10.1002/hep.29913
European Association for the Study of the Liver. Electronic address: easloffice@easloffice.eu; European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69(1):182-236. https://doi.org/10.1016/j.jhep.2018.03.019
Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67(1):358-380. https://doi.org/10.1002/hep.29086
Llovet JM, Di Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008;100(10):698-711. https://doi.org/10.1093/jnci/djn134
Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391(10127):1301-1314. https://doi.org/10.1016/S0140-6736(18)30010-2
Jha RC, Mitchell DG, Weinreb JC, Santillan CS, Yeh BM, Francois R, et al. LI-RADS categorization of benign and likely benign findings in patients at risk of hepatocellular carcinoma: a pictorial atlas. AJR Am J Roentgenol. 2014;203(1):W48-69. https://doi.org/10.2214/AJR.13.12169
Debes JD, Chan AJ, Balderramo D, Kikuchi L, Gonzalez Ballerga E, Prieto JE, et al. Hepatocellular carcinoma in South America: Evaluation of risk factors, demographics and therapy. Liver Int. 2018;38(1):136-143. https://doi.org/10.1111/liv.13502
White DL, Thrift AP, Kanwal F, Davila J, El-Serag HB. Incidence of Hepatocellular Carcinoma in All 50 United States, From 2000 Through 2012. Gastroenterology. 2017;152(4):812-820.e5. https://doi.org/10.1053/j.gastro.2016.11.020
Prieto JE, Sánchez S, Prieto RG, Rojas E, González L, Mendivelso F. Características clínicas y descompensación en pacientes con cirrosis hepática atendidos en dos centros de hepatología en la ciudad de Bogotá D.C., 2010-2014. Rev Col Gastroenterol. 2016;31(1):1-8. https://doi.org/10.22516/25007440.66
Prieto-Ortiz JE, Garzón-Orjuela N, Sánchez Pardo S, Prieto-Ortiz RG, Eslava-Schmalbach J. Sobrevida en pacientes con cirrosis de acuerdo con su etiología. Cohorte retrospectiva. Rev Colomb Gastroenterol. 2022;37(1):24-32. https://doi.org/10.22516/25007440.703
Marín-Zuluaga JI, Vergara-Cadavid J, Cajiao-Castro L, Arroyave-Zuluaga D, Castro-Sánchez S, Ceballos-Ramírez L, et al. Caracterización, manejo y pronóstico de una cohorte de pacientes cirróticos con carcinoma hepatocelular. Hospital Pablo Tobón Uribe 2012-2018. Hepatología. 2020;1(2):134-44.
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64(1):73-84. https://doi.org/10.1002/hep.28431
Singal AG, Lampertico P, Nahon P. Epidemiology and surveillance for hepatocellular carcinoma: New trends. J Hepatol. 2020;72(2):250-261. https://doi.org/10.1016/j.jhep.2019.08.025
Ascha MS, Hanouneh IA, Lopez R, Tamimi TA, Feldstein AF, Zein NN. The incidence and risk factors of hepatocellular carcinoma in patients with nonalcoholic steatohepatitis. Hepatology. 2010;51(6):1972-8. https://doi.org/10.1002/hep.23527
Kanwal F, Kramer JR, Mapakshi S, Natarajan Y, Chayanupatkul M, Richardson PA, et al. Risk of Hepatocellular Cancer in Patients With Non-Alcoholic Fatty Liver Disease. Gastroenterology. 2018;155(6):1828-1837.e2. https://doi.org/10.1053/j.gastro.2018.08.024
A new prognostic system for hepatocellular carcinoma: a retrospective study of 435 patients: the Cancer of the Liver Italian Program (CLIP) investigators. Hepatology. 1998;28(3):751-5. https://doi.org/10.1002/hep.510280322
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359(4):378-90. https://doi.org/10.1056/NEJMoa0708857
Bruix J, Qin S, Merle P, Granito A, Huang YH, Bodoky G, et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;389(10064):56-66. https://doi.org/10.1016/S0140-6736(16)32453-9
Leal CRG, Magalhães C, Barbosa D, Aquino D, Carvalho B, Balbi E, et al. Survival and tolerance to sorafenib in Child-Pugh B patients with hepatocellular carcinoma: a prospective study. Invest New Drugs. 2018;36(5):911-918. https://doi.org/10.1007/s10637-018-0621-x
McNamara MG, Slagter AE, Nuttall C, Frizziero M, Pihlak R, Lamarca A, et al. Sorafenib as first-line therapy in patients with advanced Child-Pugh B hepatocellular carcinoma-a meta-analysis. Eur J Cancer. 2018;105:1-9. https://doi.org/10.1016/j.ejca.2018.09.031
Iavarone M, Primignani M, Vavassori S, Sangiovanni A, La Mura V, Romeo R, et al. Determinants of esophageal varices bleeding in patients with advanced hepatocellular carcinoma treated with sorafenib. United European Gastroenterol J. 2016;4(3):363-70. https://doi.org/10.1177/2050640615615041
Dai C, Zhou F, Shao JH, Wu LQ, Yu X, Yin XB. Bleeding risk in cancer patients treated with sorafenib: A meta-analysis of randomized controlled trials. J Cancer Res Ther. 2018;14(Supplement):S948-S956. https://doi.org/10.4103/0973-1482.188430
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Revista colombiana de Gastroenterología

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Aquellos autores/as que tengan publicaciones con esta revista, aceptan los términos siguientes:
Los autores/as ceden sus derechos de autor y garantizarán a la revista el derecho de primera publicación de su obra, el cuál estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons que permite a terceros compartir la obra siempre que se indique su autor y su primera publicación en esta revista.
Los contenidos están protegidos bajo una licencia de Creative Commons Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional.

| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |