Aspectos clave en la vacunación contra la COVID-19 en pacientes con enfermedad inflamatoria intestinal
DOI:
https://doi.org/10.22516/25007440.785Palabras clave:
COVID-19, Vacunas COVID-19, SARS-CoV-2, Vacunación, Inflamación intestinalResumen
Actualmente, la pandemia por la enfermedad por coronavirus de 2019 (COVID-19) ha sido uno de los grandes retos para el personal de la salud y se ha convertido en un mayor desafío cuando hay pacientes con enfermedad inflamatoria intestinal (EII); debido a la fisiopatología de la enfermedad y los tratamientos utilizados, se pueden generar interrogantes difíciles de contestar ante el corto tiempo de evidencia científica existente sobre la vacunación para coronavirus del síndrome respiratorio agudo grave de tipo 2 (SARS-CoV-2). Es claro que la vacunación es una de las intervenciones de salud pública más efectivas para lograr una inmunidad colectiva en esta pandemia; por tal motivo, se realiza una revisión enfocada en los aspectos claves frente a la vacunación para la infección por SARS-CoV-2 en los pacientes con EII según la evidencia disponible.
Descargas
Referencias bibliográficas
Abraham C, Cho JH. Inflammatory bowel disease. N Engl J Med. 2009;361(21):2066-78. https://doi.org/10.1056/NEJMra0804647
Fernández-Ávila DG, Bernal-Macías S, Parra-Izquierdo V, Rincón-Riaño DN, Gutiérrez JM, Rosselli D. Prevalencia en Colombia de la enfermedad inflamatoria intestinal y el compromiso articular asociado, según información del Sistema Integral de Información de la Protección Social. Rev Colomb Reumatol. 2020;27(1):3-8. https://doi.org/10.1016/j.rcreu.2019.10.006
Saavedra Trujillo CH. Vacunación. Infectio. 2021;25(4 suppl 1):250-85. https://doi.org/10.22354/in.v25i4.978
Kirchgesner J, Lemaitre M, Carrat F, Zureik M, Carbonnel F, Dray-Spira R. Risk of Serious and Opportunistic Infections Associated With Treatment of Inflammatory Bowel Diseases. Gastroenterology. 2018;155(2):337-346.e10. https://doi.org/10.1053/j.gastro.2018.04.012
Lichtenstein GR, Feagan BG, Cohen RD, Salzberg BA, Diamond RH, Price S, et al. Serious infection and mortality in patients with Crohn's disease: more than 5 years of follow-up in the TREAT™ registry. Am J Gastroenterol. 2012;107(9):1409-22. https://doi.org/10.1038/ajg.2012.218
Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181(2):271-280.e8. https://doi.org/10.1016/j.cell.2020.02.052
Harmer D, Gilbert M, Borman R, Clark KL. Quantitative mRNA expression profiling of ACE 2, a novel homologue of angiotensin converting enzyme. FEBS Lett. 2002;532(1-2):107-10. https://doi.org/10.1016/s0014-5793(02)03640-2
Garg M, Royce SG, Tikellis C, Shallue C, Batu D, Velkoska E, et al. Imbalance of the renin-angiotensin system may contribute to inflammation and fibrosis in IBD: a novel therapeutic target? Gut. 2020;69(5):841-851. https://doi.org/10.1136/gutjnl-2019-318512
Ning L, Shan G, Sun Z, Zhang F, Xu C, Lou X, et al. Quantitative Proteomic Analysis Reveals the Deregulation of Nicotinamide Adenine Dinucleotide Metabolism and CD38 in Inflammatory Bowel Disease. Biomed Res Int. 2019;2019:3950628. https://doi.org/10.1155/2019/3950628
Ibrahim IM, Abdelmalek DH, Elshahat ME, Elfiky AA. COVID-19 spike-host cell receptor GRP78 binding site prediction. J Infect. 2020;80(5): 554-562. https://doi.org/10.21203/rs.2.24599/v1
Zhang H, Kang Z, Gong H, Xu D, Wang J, Li Z, et al. The digestive system is a potential route of 2019-nCov infection: a bioinformatics analysis based on single-cell transcriptomes. bioRxiv. 31 de enero de 2020;2020.01.30.927806. https://doi.org/10.1101/2020.01.30.927806
Kappelman M, Brenner E, Colombel JF, Ungaro R, Agrawal M. Coronavirus and IBD Reporting Database [Internet]. Secure-IBD Database [citado el 1 de abril de 2020]. Disponible en: https://covidibd.org/
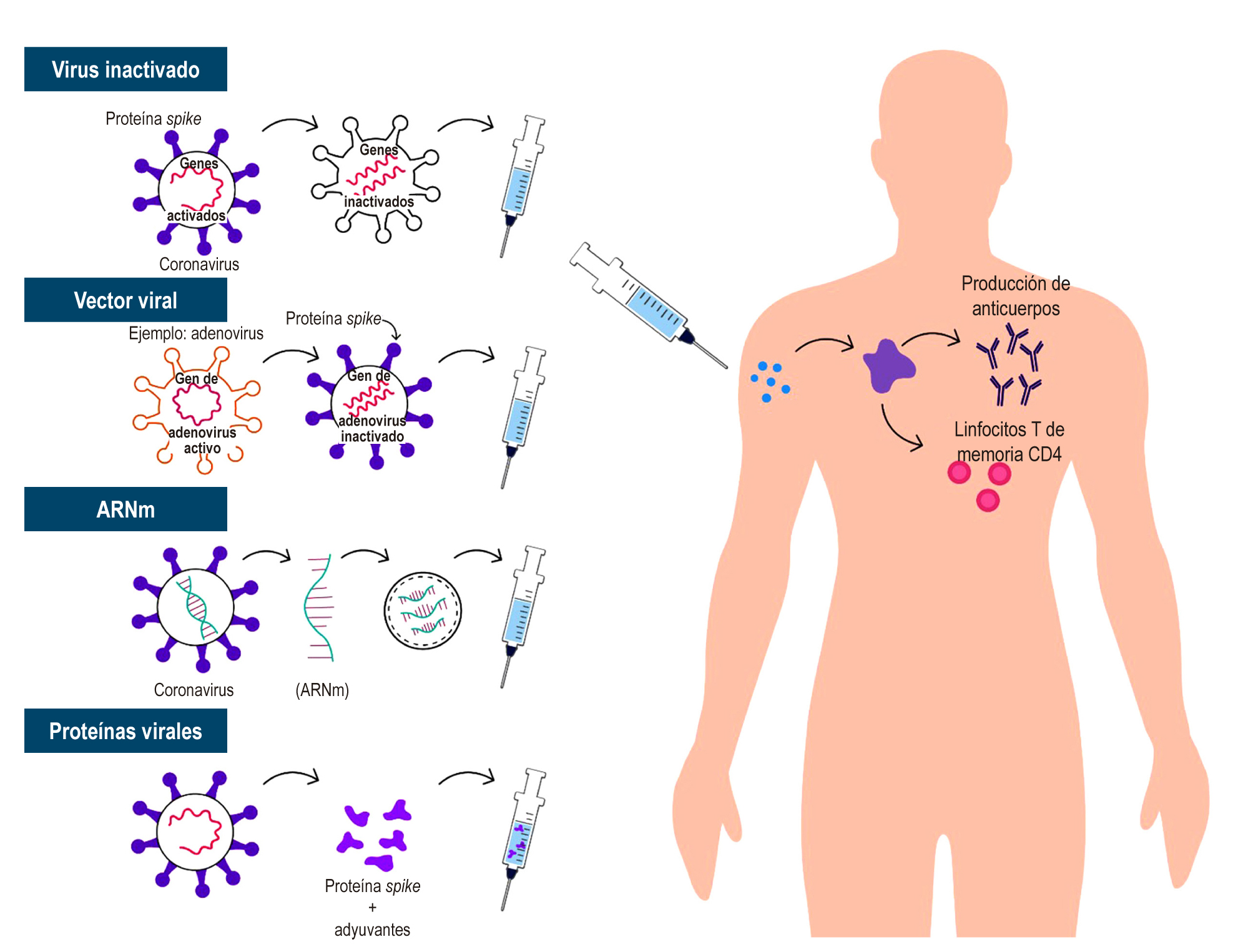
Pollard AJ, Bijker EM. A guide to vaccinology: from basic principles to new developments. Nat Rev Immunol. 2021;21(2):83-100. https://doi.org/10.1038/s41577-020-00479-7
Draft landscape and tracker of COVID-19 candidate vaccines [Internet]. World Health Organization [citado el 20 de mayo de 2021]. Disponible en: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines
Thanh Le T, Andreadakis Z, Kumar A, Gómez Román R, Tollefsen S, Saville M, et al. The COVID-19 vaccine development landscape. Nat Rev Drug Discov. 2020;19(5):305-306. https://doi.org/10.1038/d41573-020-00073-5
Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, Perez JL, et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. 2020;383(27):2603-2615. https://doi.org/10.1056/NEJMoa2034577
Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, Novak R, et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. 2021;384(5):403-416. https://doi.org/10.1056/NEJMoa2035389
Sadoff J, Gray G, Vandebosch A, Cárdenas V, Shukarev G, Grinsztejn B, et al. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against Covid-19. N Engl J Med. 2021. https://doi.org/10.1056/NEJMoa2101544
Logunov DY, Dolzhikova IV, Shcheblyakov DV, Tukhvatulin AI, Zubkova OV, Dzharullaeva AS, et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet. 2021;397(10275):671-681. https://doi.org/10.1016/S0140-6736(21)00234-8
Voysey M, Clemens SAC, Madhi SA, Weckx LY, Folegatti PM, Aley PK, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397(10269):99-111. https://doi.org/10.1016/S0140-6736(20)32661-1
Zhang Y, Zeng G, Pan H, Li C, Hu Y, Chu K, et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18-59 years: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect Dis. 2021;21(2):181-192. https://doi.org/10.1016/S1473-3099(20)30843-4
Wu Z, Hu Y, Xu M, Chen Z, Yang W, Jiang Z, et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy adults aged 60 years and older: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect Dis. 2021;21(6):803-812. https://doi.org/10.1016/S1473-3099(20)30987-7
Principles of Vaccination. En: Epidemiology and Prevention of Vaccine-Preventable Diseases [Internet]. 13.a edición. CDC; 2015 [actualizado el 29 de junio de 2020; citado el 20 de mayo de 2021]. Disponible en: https://www.cdc.gov/vaccines/pubs/pinkbook/prinvac.html
Voysey M, Costa Clemens SA, Madhi SA, Weckx LY, Folegatti PM, Aley PK, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. 2021;397(10269):99-11. https://doi.org/10.1016/ S0140-6736(20)32661-1
Vartak A, Sucheck SJ. Recent Advances in Subunit Vaccine Carriers. Vaccines (Basel). 2016;4(2):12. https://doi.org/10.3390/vaccines4020012
Rubin DT, Abreu MT, Rai V, Siegel CA; International Organization for the Study of Inflammatory Bowel Disease. Management of Patients With Crohn's Disease and Ulcerative Colitis During the Coronavirus Disease-2019 Pandemic: Results of an International Meeting. Gastroenterology. 2020;159(1):6-13.e6. https://doi.org/10.1053/j.gastro.2020.04.002
Siegel CA, Melmed GY, McGovern DP, Rai V, Krammer F, Rubin DT, et al. SARS-CoV-2 vaccination for patients with inflammatory bowel diseases: recommendations from an international consensus meeting. Gut. 2021;70(4):635-640. https://doi.org/10.1136/gutjnl-2020-324000
Alexander JL, Moran GW, Gaya DR, Raine T, Hart A, Kennedy NA, et al. SARS-CoV-2 vaccination for patients with inflammatory bowel disease: a British Society of Gastroenterology Inflammatory Bowel Disease section and IBD Clinical Research Group position statement. Lancet Gastroenterol Hepatol. 2021;6(3):218-224. https://doi.org/10.1016/S2468-1253(21)00024-8
D'Amico F, Rabaud C, Peyrin-Biroulet L, Danese S. SARS-CoV-2 vaccination in IBD: more pros than cons. Nat Rev Gastroenterol Hepatol. 2021;18(4):211-213. https://doi.org/10.1038/s41575-021-00420-w
deBruyn J, Fonseca K, Ghosh S, Panaccione R, Gasia MF, Ueno A, et al. Immunogenicity of Influenza Vaccine for Patients with Inflammatory Bowel Disease on Maintenance Infliximab Therapy: A Randomized Trial. Inflamm Bowel Dis. 2016;22(3):638-47. https://doi.org/10.1097/MIB.0000000000000615
Melmed GY, Agarwal N, Frenck RW, Ippoliti AF, Ibanez P, Papadakis KA, et al. Immunosuppression impairs response to pneumococcal polysaccharide vaccination in patients with inflammatory bowel disease. Am J Gastroenterol. 2010;105(1):148-54. https://doi.org/10.1038/ajg.2009.523
Pratt PK Jr, David N, Weber HC, Little FF, Kourkoumpetis T, Patts GJ, et al. Antibody Response to Hepatitis B Virus Vaccine is Impaired in Patients With Inflammatory Bowel Disease on Infliximab Therapy. Inflamm Bowel Dis. 2018;24(2):380-386. https://doi.org/10.1093/ibd/izx001
Agarwal N, Ollington K, Kaneshiro M, Frenck R, Melmed GY. Are immunosuppressive medications associated with decreased responses to routine immunizations? A systematic review. Vaccine. 2012;30(8):1413-24. https://doi.org/10.1016/j.vaccine.2011.11.109
Winthrop KL, Silverfield J, Racewicz A, Neal J, Lee EB, Hrycaj P, et al. The effect of tofacitinib on pneumococcal and influenza vaccine responses in rheumatoid arthritis. Ann Rheum Dis. 2016;75(4):687-95. https://doi.org/10.1136/annrheumdis-2014-207191
Doornekamp L, Goetgebuer RL, Schmitz KS, Goeijenbier M, van der Woude CJ, Fouchier R, et al. High Immunogenicity to Influenza Vaccination in Crohn's Disease Patients Treated with Ustekinumab. Vaccines (Basel). 2020;8(3):455. https://doi.org/10.3390/vaccines8030455
Brodmerkel C, Wadman E, Langley RG, Papp KA, Bourcier M, Poulin Y, et al. Immune response to pneumococcus and tetanus toxoid in patients with moderate-to-severe psoriasis following long-term ustekinumab use. J Drugs Dermatol. 2013;12(10):1122-9.
Caldera F, Hillman L, Saha S, Wald A, Grimes I, Zhang Y, et al. Immunogenicity of High Dose Influenza Vaccine for Patients with Inflammatory Bowel Disease on Anti-TNF Monotherapy: A Randomized Clinical Trial. Inflamm Bowel Dis. 2020;26(4):593-602. https://doi.org/10.1093/ibd/izz164
Wyant T, Leach T, Sankoh S, Wang Y, Paolino J, Pasetti MF, et al. Vedolizumab affects antibody responses to immunisation selectively in the gastrointestinal tract: randomised controlled trial results. Gut. 2015;64(1):77-83. https://doi.org/10.1136/gutjnl-2014-307127
Flanagan KL, Fink AL, Plebanski M, Klein SL. Sex and Gender Differences in the Outcomes of Vaccination over the Life Course. Annu Rev Cell Dev Biol. 2017;33:577-599. https://doi.org/10.1146/annurev-cellbio-100616-060718
Sheridan PA, Paich HA, Handy J, Karlsson EA, Hudgens MG, Sammon AB, et al. Obesity is associated with impaired immune response to influenza vaccination in humans. Int J Obes (Lond). 2012;36(8):1072-7. https://doi.org/10.1038/ijo.2011.208
Dhakal S, Klein SL. Host Factors Impact Vaccine Efficacy: Implications for Seasonal and Universal Influenza Vaccine Programs. J Virol. 2019;93(21):e00797-19. https://doi.org/10.1128/JVI.00797-19
Campos LM, Silva CA, Aikawa NE, Jesus AA, Moraes JC, Miraglia J, et al. High disease activity: an independent factor for reduced immunogenicity of the pandemic influenza a vaccine in patients with juvenile systemic lupus erythematosus. Arthritis Care Res (Hoboken). 2013;65(7):1121-7. https://doi.org/10.1002/acr.21948
Ellington S, Strid P, Tong VT, Woodworth K, Galang RR, Zambrano LD, et al. Characteristics of Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status - United States, January 22-June 7, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(25):769-775. https://doi.org/10.15585/mmwr.mm6925a1
Vera-Lastra O, Medina G, Cruz-Dominguez Mdel P, Jara LJ, Shoenfeld Y. Autoimmune/inflammatory syndrome induced by adjuvants (Shoenfeld's syndrome): clinical and immunological spectrum. Expert Rev Clin Immunol. 2013;9(4):361-73. https://doi.org/10.1586/eci.13.2
Münz C, Lünemann JD, Getts MT, Miller SD. Antiviral immune responses: triggers of or triggered by autoimmunity? Nat Rev Immunol. 2009;9(4):246-58. https://doi.org/10.1038/nri2527
Agmon-Levin N, Paz Z, Israeli E, Shoenfeld Y. Vaccines and autoimmunity. Nat Rev Rheumatol. 2009;5(11):648-52. https://doi.org/10.1038/nrrheum.2009.196
Schattner A. Consequence or coincidence? The occurrence, pathogenesis and significance of autoimmune manifestations after viral vaccines. Vaccine. 2005;23(30):3876-86. https://doi.org/10.1016/j.vaccine.2005.03.005
Salemi S, D'Amelio R. Could autoimmunity be induced by vaccination? Int Rev Immunol. 2010;29(3):247-69. https://doi.org/10.3109/08830181003746304
Vadalà M, Poddighe D, Laurino C, Palmieri B. Vaccination and autoimmune diseases: is prevention of adverse health effects on the horizon? EPMA J. 2017;8(3):295-311. https://doi.org/10.1007/s13167-017-0101-y
De Martino M, Chiappini E, Galli L. Vaccines and autoimmunity. Int J Immunopathol Pharmacol. 2013;26(2):283-90. https://doi.org/10.1177/039463201302600201
Rodríguez Y, Rojas M, Pacheco Y, Acosta-Ampudia Y, Ramírez-Santana C, Monsalve DM, et al. Guillain-Barré syndrome, transverse myelitis and infectious diseases. Cell Mol Immunol. 2018;15(6):547-562. https://doi.org/10.1038/cmi.2017.142
Markovic-Plese S, Hemmer B, Zhao Y, Simon R, Pinilla C, Martin R. High level of cross-reactivity in influenza virus hemagglutinin-specific CD4+ T-cell response: implications for the initiation of autoimmune response in multiple sclerosis. J Neuroimmunol. 2005;169(1-2):31-8. https://doi.org/10.1016/j.jneuroim.2005.07.014
Sioofy-Khojine AB, Lehtonen J, Nurminen N, Laitinen OH, Oikarinen S, Huhtala H, et al. Coxsackievirus B1 infections are associated with the initiation of insulin-driven autoimmunity that progresses to type 1 diabetes. Diabetologia. 2018;61(5):1193-1202. https://doi.org/10.1007/s00125-018-4561-y
Takahashi Y, Murai C, Shibata S, Munakata Y, Ishii T, Ishii K, et al. Human parvovirus B19 as a causative agent for rheumatoid arthritis. Proc Natl Acad Sci U S A. 1998;95(14):8227-32. https://doi.org/10.1073/pnas.95.14.8227
Román GC, Gracia F, Torres A, Palacios A, Gracia K, Harris D. Acute Transverse Myelitis (ATM):Clinical Review of 43 Patients With COVID-19-Associated ATM and 3 Post-Vaccination ATM Serious Adverse Events With the ChAdOx1 nCoV-19 Vaccine (AZD1222). Front Immunol. 2021;12:653786. https://doi.org/10.3389/fimmu.2021.653786
Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger S. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N Engl J Med. 2021:NEJMoa2104840. https://doi.org/10.1056/NEJMoa2104840
Isaacs A, Cox RA, Rotem Z. Foreign nucleic acids as the stimulus to make interferon. Lancet. 1963;2(7299):113-6. https://doi.org/10.1016/s0140-6736(63)92585-6
Karikó K, Muramatsu H, Welsh FA, Ludwig J, Kato H, Akira S, et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol Ther. 2008;16(11):1833-40. https://doi.org/10.1038/mt.2008.200
Karikó K, Muramatsu H, Ludwig J, Weissman D. Generating the optimal mRNA for therapy: HPLC purification eliminates immune activation and improves translation of nucleoside-modified, protein-encoding mRNA. Nucleic Acids Res. 2011;39(21):e142. https://doi.org/10.1093/nar/gkr695
Saavedra Trujillo CH. Consenso colombiano de atención, diagnóstico y manejo de la infección por SARS-COV-2/COVID-19 en establecimientos de atención de la salud. Recomendaciones basadas en consenso de expertos e informadas en la evidencia. Infectio. 2020;24(3). https://doi.org/10.22354/in.v24i3.895
Velikova T, Georgiev T. SARS-CoV-2 vaccines and autoimmune diseases amidst the COVID-19 crisis. Rheumatol Int. 2021;41(3):509-518. https://doi.org/10.1007/s00296-021-04792-9
Tse F, Moayyedi P, Waschke KA, MacMillan M, Forbes N, Carroll MW, Carman N, Leontiadis GI. COVID-19 Vaccination in Patients With Inflammatory Bowel Disease: Communiqué From the Canadian Association of Gastroenterology. J Can Assoc Gastroenterol. 2021;4(1):49. https://doi.org/10.1093/jcag/gwaa046
Melmed GY, Rubin DT, McGovern DPB. Winter Is Coming! Clinical, Immunologic, and Practical Considerations for Vaccinating Patients With Inflammatory Bowel Disease During the Coronavirus Disease-2019 Pandemic. Gastroenterology. 2021;160(3):639-644. https://doi.org/10.1053/j.gastro.2020.10.013
Descargas
Publicado
Cómo citar
Número
Sección
Licencia
Aquellos autores/as que tengan publicaciones con esta revista, aceptan los términos siguientes:
Los autores/as ceden sus derechos de autor y garantizarán a la revista el derecho de primera publicación de su obra, el cuál estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons que permite a terceros compartir la obra siempre que se indique su autor y su primera publicación en esta revista.
Los contenidos están protegidos bajo una licencia de Creative Commons Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional.


| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |